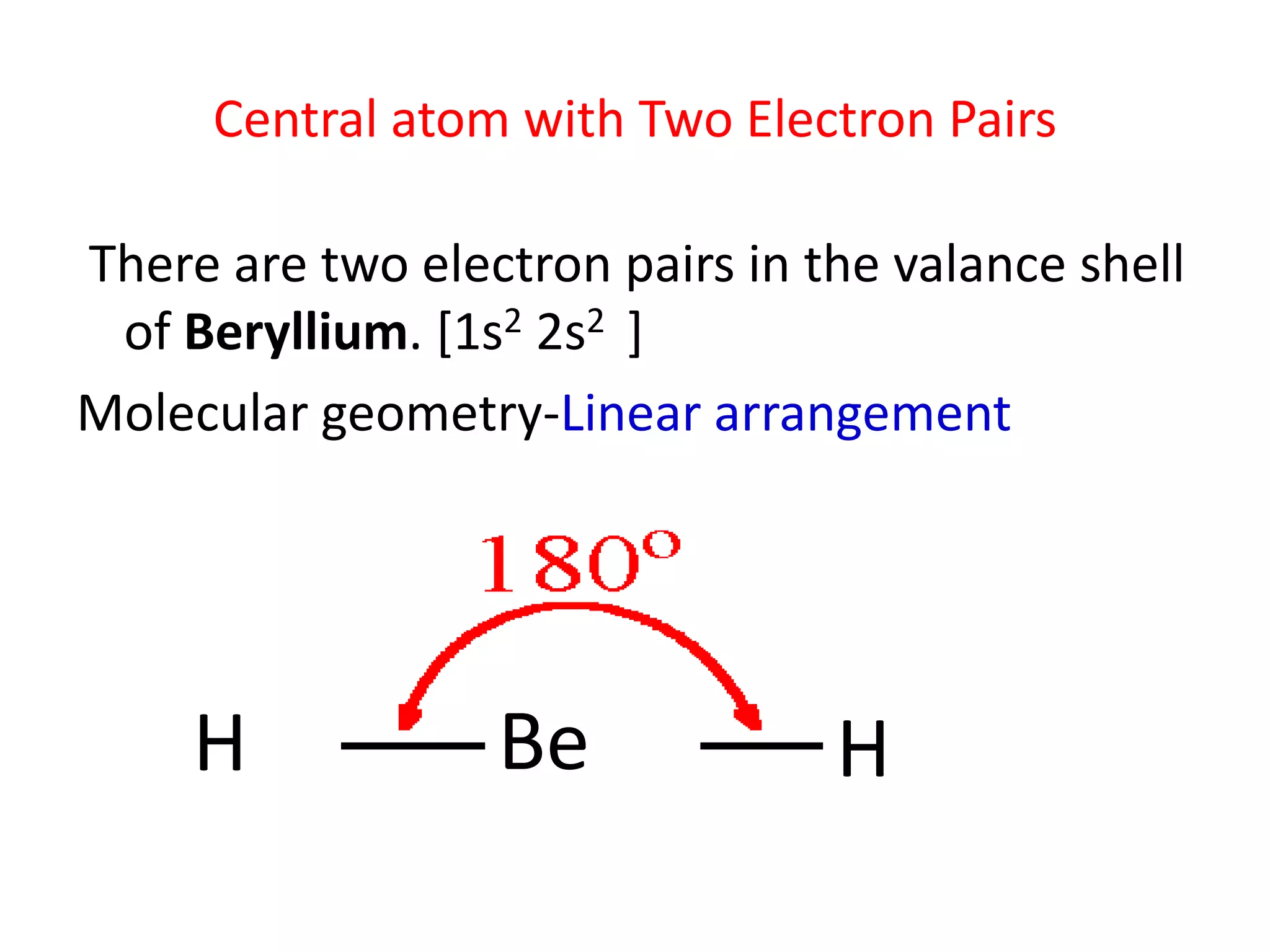
Beryllium, a chemical element with the symbol Be and atomic number 4, is a fascinating subject in the realm of chemistry. One of its most intriguing aspects is its electronic configuration, particularly the Beryllium Valence Electrons. Understanding the valence electrons of beryllium is crucial for comprehending its chemical behavior and reactivity. This post delves into the details of beryllium's valence electrons, their significance, and how they influence the element's properties and applications.
Understanding Valence Electrons
Valence electrons are the electrons in the outermost shell of an atom. These electrons are responsible for the chemical properties of an element, including its reactivity and bonding behavior. The number of valence electrons determines how an atom will interact with other atoms to form compounds.
The Electronic Configuration of Beryllium
Beryllium has an atomic number of 4, which means it has 4 protons and 4 electrons. The electronic configuration of beryllium is 1s22s2. This configuration indicates that beryllium has two electrons in the first shell (1s2) and two electrons in the second shell (2s2). The two electrons in the second shell are the Beryllium Valence Electrons.
Significance of Beryllium Valence Electrons
The presence of two valence electrons in beryllium has several important implications:
- Chemical Reactivity: Beryllium is relatively non-reactive compared to other alkaline earth metals. This is because it has a stable electronic configuration with a full s-subshell in its outermost shell. The two valence electrons are tightly bound to the nucleus, making it less likely to lose or gain electrons.
- Bonding Behavior: Despite its low reactivity, beryllium can form covalent bonds with other elements. The two valence electrons can participate in bonding, leading to the formation of compounds such as beryllium chloride (BeCl2) and beryllium oxide (BeO).
- Physical Properties: The electronic configuration of beryllium contributes to its unique physical properties. For example, beryllium has a high melting point and is a good conductor of electricity and heat. These properties make it useful in various industrial applications.
Applications of Beryllium
Beryllium’s unique properties, influenced by its Beryllium Valence Electrons, make it valuable in several industries:
- Aerospace Industry: Beryllium is used in the aerospace industry due to its lightweight and high strength. It is often alloyed with other metals to create materials that can withstand extreme conditions.
- Electronics: Beryllium is used in the production of electronic components, such as transistors and diodes, because of its excellent thermal conductivity.
- Medical Equipment: Beryllium is used in medical imaging equipment, such as X-ray machines, due to its ability to absorb X-rays.
- Nuclear Reactors: Beryllium is used as a neutron reflector in nuclear reactors. Its ability to reflect neutrons helps in controlling the nuclear reaction.
Chemical Compounds of Beryllium
Beryllium forms various compounds, primarily through the involvement of its valence electrons. Some of the notable compounds include:
- Beryllium Oxide (BeO): This compound is used in ceramics and as a refractory material due to its high melting point and thermal conductivity.
- Beryllium Chloride (BeCl2): This compound is used in the production of other beryllium compounds and as a catalyst in various chemical reactions.
- Beryllium Fluoride (BeF2): This compound is used in the production of optical materials and as a flux in metallurgy.
Safety Considerations
While beryllium has many useful applications, it is also highly toxic. Inhalation of beryllium dust or fumes can lead to a condition known as berylliosis, a chronic lung disease. Therefore, handling beryllium requires strict safety measures, including the use of personal protective equipment and proper ventilation.
🛑 Note: Always follow safety guidelines when working with beryllium to avoid health risks.
Environmental Impact
Beryllium mining and processing can have significant environmental impacts. The extraction of beryllium often involves the use of hazardous chemicals, which can contaminate soil and water. Additionally, the disposal of beryllium waste requires careful management to prevent environmental pollution.
🌿 Note: Proper waste management practices are essential to mitigate the environmental impact of beryllium mining and processing.
Future Prospects
The unique properties of beryllium, driven by its Beryllium Valence Electrons, continue to make it a valuable material in various industries. As technology advances, new applications for beryllium are likely to emerge, further enhancing its importance. However, the challenges associated with its toxicity and environmental impact must be addressed to ensure sustainable use.
Research is ongoing to develop safer and more efficient methods for extracting and processing beryllium. Additionally, efforts are being made to find alternative materials that can replace beryllium in certain applications, reducing the reliance on this toxic element.
In conclusion, the Beryllium Valence Electrons play a crucial role in determining the element’s chemical and physical properties. Understanding these electrons is essential for appreciating beryllium’s unique characteristics and its wide range of applications. While beryllium offers numerous benefits, its toxicity and environmental impact must be carefully managed to ensure safe and sustainable use. As research continues, the future of beryllium in various industries looks promising, with ongoing efforts to address its challenges and explore new possibilities.
Related Terms:
- beryllium valence shell electrons
- beryllium electron configuration
- boron how many valence electrons
- what is valence electron configuration
- electron arrangement of beryllium
- aufbau diagram for beryllium