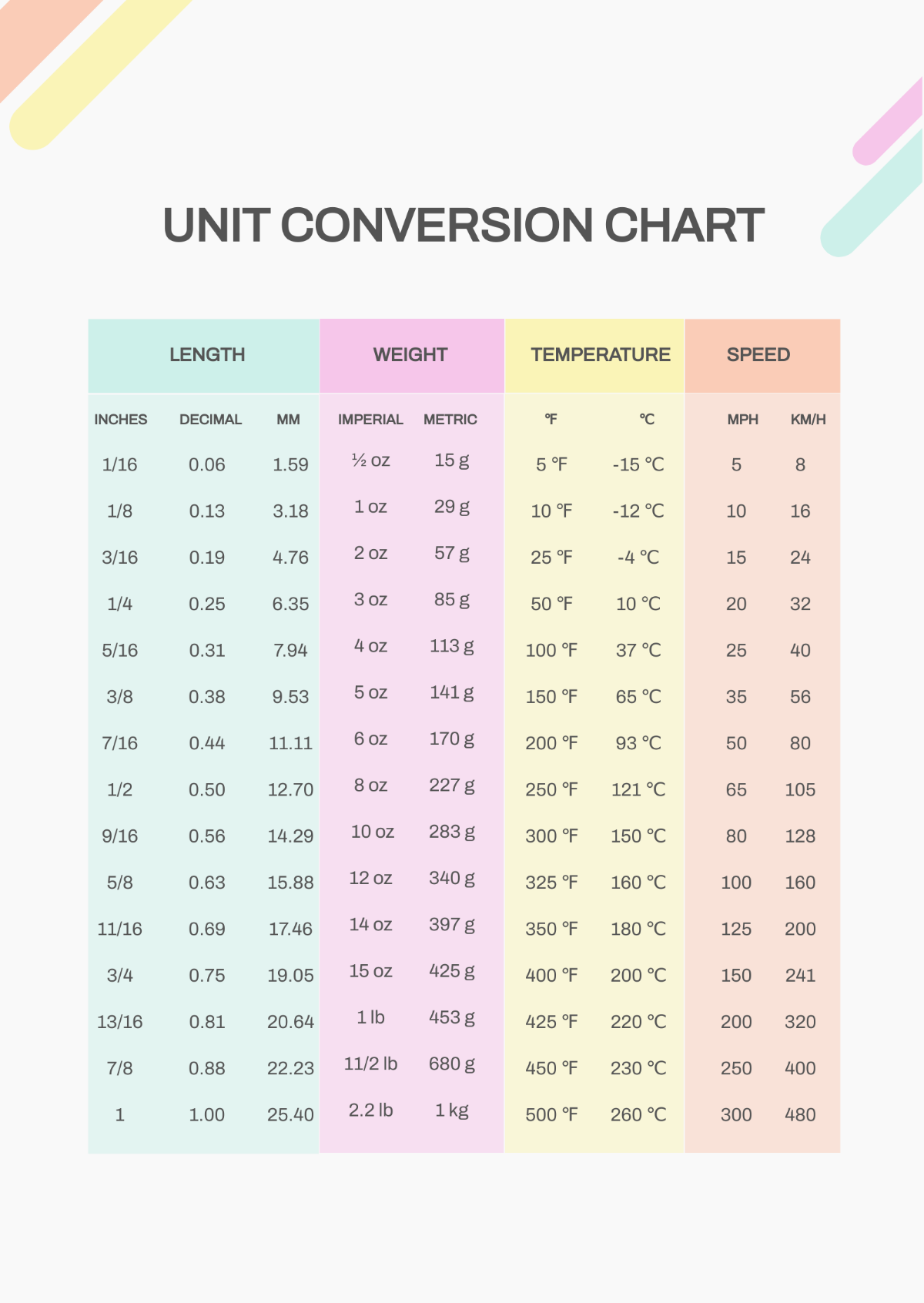
Mastering chem unit conversions is a fundamental skill for anyone studying chemistry or related sciences. Whether you're a student preparing for exams or a professional working in a lab, understanding how to convert between different units of measurement is crucial. This guide will walk you through the essentials of chem unit conversions, providing clear explanations and practical examples to help you become proficient.
Understanding the Basics of Chem Unit Conversions
Before diving into specific chem unit conversions, it's important to grasp the basic principles. Chemistry often involves measurements in various units, such as grams, moles, liters, and meters. Converting between these units requires a solid understanding of conversion factors and dimensional analysis.
Conversion factors are ratios that relate two different units of measurement. For example, the conversion factor between grams and kilograms is 1 kg = 1000 g. This means that 1 kilogram is equal to 1000 grams. Conversion factors are used to cancel out unwanted units and leave the desired units in your calculations.
Dimensional analysis is a method used to convert units by multiplying the quantity by one or more conversion factors. This method ensures that the units cancel out correctly, leaving you with the desired unit. For example, to convert 500 grams to kilograms, you would use the conversion factor 1 kg / 1000 g:
500 g * (1 kg / 1000 g) = 0.5 kg
Common Units in Chemistry
Chemistry involves a variety of units, each serving a specific purpose. Some of the most common units include:
- Mass: grams (g), kilograms (kg), milligrams (mg)
- Volume: liters (L), milliliters (mL), cubic centimeters (cm³)
- Moles: moles (mol)
- Temperature: Celsius (°C), Kelvin (K)
- Pressure: atmospheres (atm), pascals (Pa), millimeters of mercury (mmHg)
Understanding these units and how to convert between them is essential for accurate chem unit conversions.
Converting Between Mass Units
Mass is a fundamental property in chemistry, and converting between different mass units is a common task. Here are some examples of mass unit conversions:
- Grams to kilograms: 1 kg = 1000 g
- Grams to milligrams: 1 g = 1000 mg
- Kilograms to grams: 1 kg = 1000 g
For example, to convert 2.5 kilograms to grams:
2.5 kg * (1000 g / 1 kg) = 2500 g
To convert 350 milligrams to grams:
350 mg * (1 g / 1000 mg) = 0.35 g
Converting Between Volume Units
Volume is another crucial measurement in chemistry, often used to describe the amount of a substance. Common volume units include liters, milliliters, and cubic centimeters. Here are some examples of volume unit conversions:
- Liters to milliliters: 1 L = 1000 mL
- Milliliters to cubic centimeters: 1 mL = 1 cm³
- Liters to cubic centimeters: 1 L = 1000 cm³
For example, to convert 4.5 liters to milliliters:
4.5 L * (1000 mL / 1 L) = 4500 mL
To convert 750 cubic centimeters to milliliters:
750 cm³ * (1 mL / 1 cm³) = 750 mL
Converting Between Moles and Mass
Moles are a fundamental unit in chemistry, representing the amount of a substance. Converting between moles and mass requires knowledge of the substance's molar mass. The molar mass is the mass of one mole of a substance and is typically expressed in grams per mole (g/mol).
To convert moles to mass, use the formula:
Mass (g) = Moles (mol) * Molar Mass (g/mol)
For example, to convert 2 moles of water (H₂O) to grams, knowing that the molar mass of water is approximately 18.015 g/mol:
Mass = 2 mol * 18.015 g/mol = 36.03 g
To convert mass to moles, use the formula:
Moles (mol) = Mass (g) / Molar Mass (g/mol)
For example, to convert 50 grams of glucose (C₆H₁₂O₆) to moles, knowing that the molar mass of glucose is approximately 180.156 g/mol:
Moles = 50 g / 180.156 g/mol ≈ 0.278 mol
Converting Between Temperature Units
Temperature is a critical measurement in many chemical reactions. The most common temperature units in chemistry are Celsius and Kelvin. Converting between these units is straightforward:
- Celsius to Kelvin: K = °C + 273.15
- Kelvin to Celsius: °C = K - 273.15
For example, to convert 25°C to Kelvin:
K = 25°C + 273.15 = 298.15 K
To convert 300 K to Celsius:
°C = 300 K - 273.15 = 26.85°C
Converting Between Pressure Units
Pressure is another important measurement in chemistry, often used to describe the conditions of a reaction. Common pressure units include atmospheres, pascals, and millimeters of mercury. Here are some examples of pressure unit conversions:
- Atmospheres to pascals: 1 atm = 101325 Pa
- Millimeters of mercury to atmospheres: 1 mmHg = 0.00131579 atm
- Pascals to atmospheres: 1 Pa = 9.86923 × 10⁻⁶ atm
For example, to convert 760 mmHg to atmospheres:
760 mmHg * (0.00131579 atm / 1 mmHg) = 1 atm
To convert 101325 Pa to atmospheres:
101325 Pa * (1 atm / 101325 Pa) = 1 atm
Practical Examples of Chem Unit Conversions
Let's go through some practical examples to solidify your understanding of chem unit conversions.
Example 1: Convert 3.5 liters of water to milliliters.
3.5 L * (1000 mL / 1 L) = 3500 mL
Example 2: Convert 500 grams of sucrose (C₁₂H₂₂O₁₁) to moles, knowing that the molar mass of sucrose is approximately 342.3 g/mol.
Moles = 500 g / 342.3 g/mol ≈ 1.46 mol
Example 3: Convert 250 milliliters of a solution to liters.
250 mL * (1 L / 1000 mL) = 0.25 L
Example 4: Convert 300 Kelvin to Celsius.
°C = 300 K - 273.15 = 26.85°C
Example 5: Convert 1.5 atmospheres to pascals.
1.5 atm * (101325 Pa / 1 atm) = 151987.5 Pa
📝 Note: Always double-check your conversion factors and calculations to ensure accuracy.
Common Mistakes to Avoid in Chem Unit Conversions
Even with a solid understanding of chem unit conversions, it's easy to make mistakes. Here are some common pitfalls to avoid:
- Incorrect Conversion Factors: Ensure that your conversion factors are accurate and relevant to the units you are converting.
- Unit Cancellation Errors: Double-check that your units cancel out correctly, leaving you with the desired unit.
- Rounding Errors: Be mindful of significant figures and round your answers appropriately.
- Mistaken Molar Masses: Verify the molar mass of the substance you are working with to avoid errors in mole-to-mass conversions.
By being aware of these common mistakes, you can improve the accuracy of your chem unit conversions and avoid costly errors in your calculations.
Advanced Chem Unit Conversions
Once you are comfortable with the basics of chem unit conversions, you can explore more advanced topics. These include converting between different systems of units, such as the metric system and the imperial system, and handling more complex chemical calculations.
For example, converting between metric and imperial units:
- Grams to ounces: 1 oz = 28.3495 g
- Liters to gallons: 1 gal = 3.78541 L
- Meters to feet: 1 ft = 0.3048 m
For example, to convert 500 grams to ounces:
500 g * (1 oz / 28.3495 g) ≈ 17.637 oz
To convert 2 liters to gallons:
2 L * (1 gal / 3.78541 L) ≈ 0.528 gal
To convert 10 meters to feet:
10 m * (1 ft / 0.3048 m) ≈ 32.808 ft
Advanced chem unit conversions often involve more complex calculations and may require additional knowledge of chemistry principles. However, with practice and a solid foundation in the basics, you can master these advanced topics.
Conclusion
Mastering chem unit conversions is essential for anyone studying or working in chemistry. By understanding the basic principles of conversion factors and dimensional analysis, you can accurately convert between different units of measurement. Whether you’re converting mass, volume, moles, temperature, or pressure, the key is to practice and double-check your calculations. With the knowledge and examples provided in this guide, you should be well on your way to becoming proficient in chem unit conversions.
Related Terms:
- important conversions in chemistry
- understanding conversions in chemistry
- conversion in chemistry
- all conversions in chemistry
- gen chem unit conversions
- general chemistry conversions