Understanding the electron configuration of Cs is fundamental to grasping the chemical behavior and properties of cesium, an alkali metal with the atomic number 55. Cesium is known for its highly reactive nature and its use in various applications, including atomic clocks and photoelectric cells. This blog post delves into the electron configuration of cesium, its significance, and how it influences the element's properties and applications.
What is Electron Configuration?
Electron configuration refers to the arrangement of electrons in the orbitals of an atom. It is a crucial concept in chemistry as it helps predict the chemical behavior of elements. The configuration is typically written using a notation that indicates the energy levels (shells) and sublevels (orbitals) where the electrons are located.
Electron Configuration of Cesium
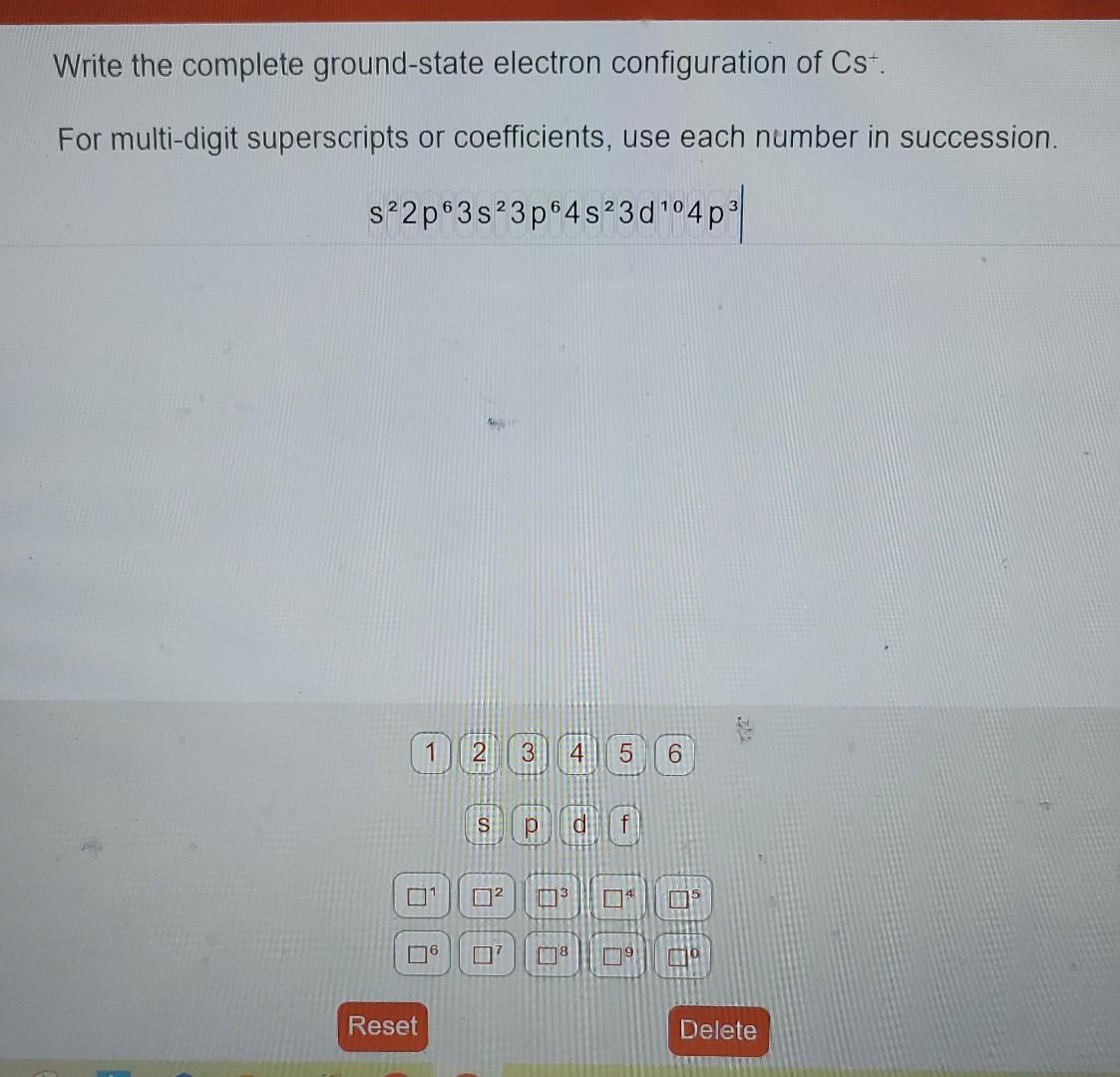
The electron configuration of Cs can be determined by following the Aufbau principle, which states that electrons fill the lowest energy levels first before moving to higher energy levels. Cesium has 55 electrons, and its electron configuration is:
1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 5s2 5p6 6s1
This configuration can be broken down as follows:
- 1s2: The first shell contains 2 electrons.
- 2s2 2p6: The second shell contains 8 electrons.
- 3s2 3p6 3d10: The third shell contains 18 electrons.
- 4s2 4p6 4d10: The fourth shell contains 18 electrons.
- 5s2 5p6: The fifth shell contains 8 electrons.
- 6s1: The sixth shell contains 1 electron.
This configuration shows that cesium has a single electron in its outermost shell, which is characteristic of alkali metals. This single electron in the 6s orbital is responsible for cesium’s high reactivity and its tendency to lose an electron to form a positive ion (Cs+).
Significance of the Electron Configuration of Cs
The electron configuration of Cs has several significant implications for its chemical and physical properties:
- High Reactivity: The single electron in the outermost shell makes cesium highly reactive. It readily loses this electron to form a stable noble gas configuration, which is why cesium is often found in the +1 oxidation state.
- Low Ionization Energy: Cesium has one of the lowest ionization energies among all elements. This means it requires less energy to remove the outermost electron, making it highly reactive.
- Low Melting and Boiling Points: Despite its high reactivity, cesium has a relatively low melting point (28.5°C) and boiling point (671°C). This is due to the weak metallic bonding between cesium atoms.
- High Density: Cesium is one of the densest metals, with a density of 1.873 g/cm3 at room temperature. This is due to its large atomic size and the efficient packing of its atoms.
Applications of Cesium
The unique properties of cesium, stemming from its electron configuration of Cs, make it valuable in various applications:
- Atomic Clocks: Cesium is used in atomic clocks, which are the most accurate timekeeping devices. The cesium-133 atom is used as a reference for the second in the International System of Units (SI).
- Photoelectric Cells: Cesium is used in photoelectric cells due to its low work function, which allows it to emit electrons easily when exposed to light.
- Drilling Fluids: Cesium formate is used in drilling fluids for oil and gas exploration. Its high density helps to balance the pressure in deep wells.
- Medical Imaging: Cesium-137 is used in medical imaging and radiation therapy due to its gamma radiation properties.
Electron Configuration and Periodic Trends
The electron configuration of Cs also helps explain its position in the periodic table and its relationship with other elements. Cesium is an alkali metal, which means it is in Group 1 of the periodic table. Alkali metals share similar properties due to their electron configuration, which includes a single electron in the outermost shell.
As you move down Group 1, the atomic size increases, and the ionization energy decreases. This trend is evident in cesium, which has a larger atomic size and lower ionization energy compared to lighter alkali metals like lithium and sodium.
Here is a table showing the electron configurations of some alkali metals:
| Element | Atomic Number | Electron Configuration |
|---|---|---|
| Lithium (Li) | 3 | 1s2 2s1 |
| Sodium (Na) | 11 | 1s2 2s2 2p6 3s1 |
| Potassium (K) | 19 | 1s2 2s2 2p6 3s2 3p6 4s1 |
| Rubidium (Rb) | 37 | 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 5s1 |
| Cesium (Cs) | 55 | 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 5s2 5p6 6s1 |
📝 Note: The electron configuration of cesium follows the pattern of alkali metals, with a single electron in the outermost shell, which is characteristic of Group 1 elements.
Chemical Reactions of Cesium
The electron configuration of Cs influences its chemical reactions. Cesium readily reacts with water, oxygen, and halogens due to its high reactivity. Some common reactions include:
- Reaction with Water: Cesium reacts violently with water to form cesium hydroxide (CsOH) and hydrogen gas (H2). The reaction is exothermic and produces a lot of heat.
- Reaction with Oxygen: Cesium reacts with oxygen to form cesium oxide (Cs2O) or cesium peroxide (Cs2O2), depending on the conditions.
- Reaction with Halogens: Cesium reacts with halogens (fluorine, chlorine, bromine, iodine) to form cesium halides (CsF, CsCl, CsBr, CsI). These reactions are highly exothermic.
Safety and Handling of Cesium
Due to its high reactivity, cesium must be handled with care. It should be stored in a dry, inert atmosphere to prevent reactions with air and moisture. When handling cesium, it is essential to use appropriate personal protective equipment (PPE) and follow safety protocols to avoid accidents.
Cesium is also radioactive, and its isotopes, such as cesium-137, are hazardous. Exposure to cesium-137 can cause radiation sickness and other health issues. Therefore, it is crucial to handle cesium isotopes with extreme caution and follow proper disposal procedures.
Cesium is a fascinating element with unique properties that make it valuable in various applications. Its electron configuration of Cs plays a crucial role in determining its chemical behavior and physical properties. Understanding the electron configuration of cesium provides insights into its reactivity, periodic trends, and applications in fields such as timekeeping, medical imaging, and drilling fluids.
Related Terms:
- cs on the periodic table
- electronic configuration of cesium
- cs element electron configuration
- full electron configuration for cs
- cs2 electron configuration
- how to calculate cesium configuration