Understanding the differences between Electron vs Molecular Geometry is crucial for anyone studying chemistry, as it helps in predicting the shapes and properties of molecules. This knowledge is fundamental in fields ranging from materials science to pharmaceuticals. By delving into the concepts of electron geometry and molecular geometry, we can better comprehend how atoms arrange themselves in space and how this arrangement influences molecular behavior.
Understanding Electron Geometry
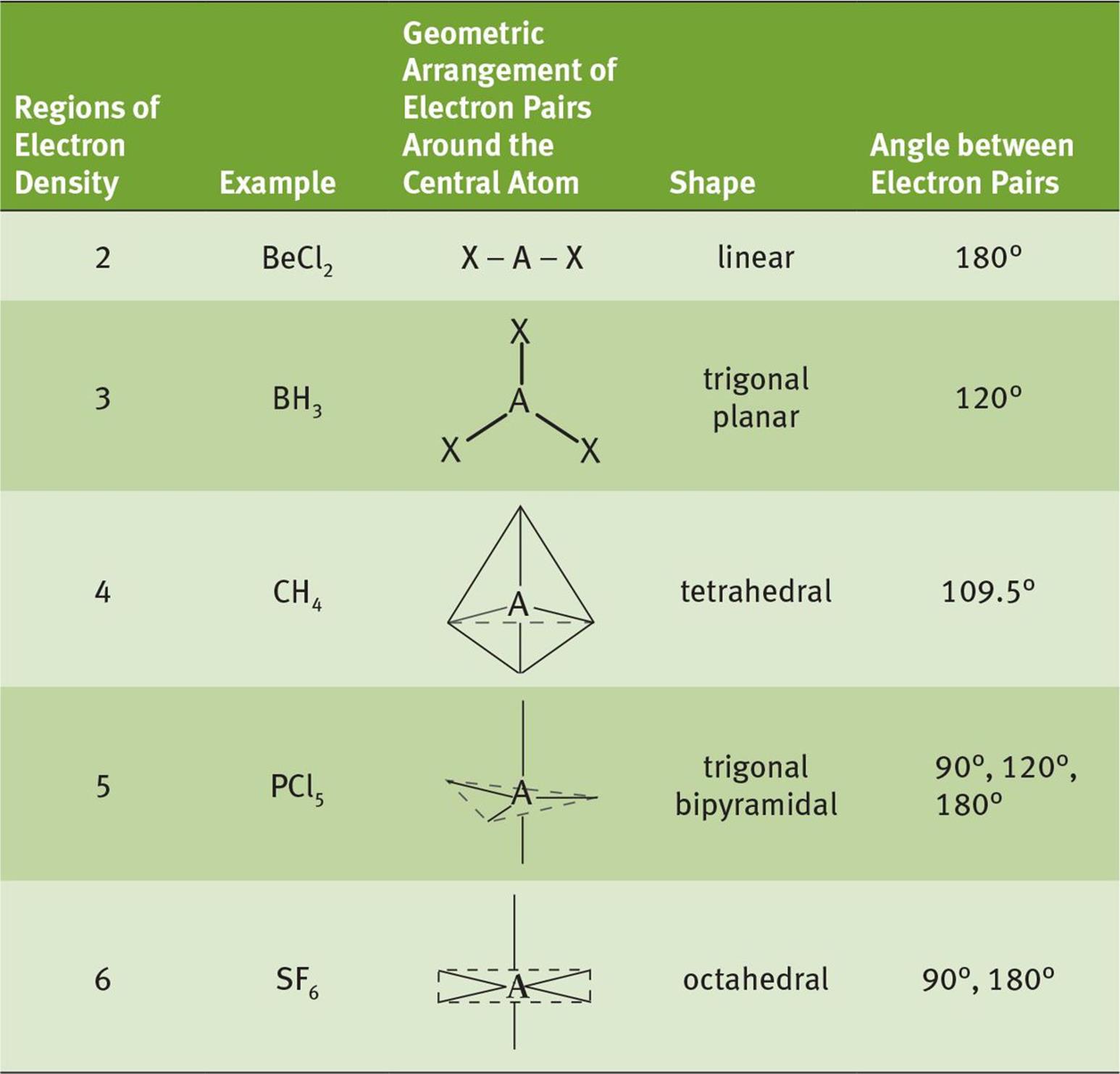
Electron geometry refers to the spatial arrangement of electron pairs around a central atom in a molecule. This arrangement is determined by the repulsion between electron pairs, which seek to minimize their mutual repulsion. The concept is based on the Valence Shell Electron Pair Repulsion (VSEPR) theory, which predicts the shapes of molecules by considering the repulsion between electron pairs in the valence shell of the central atom.
There are several key shapes in electron geometry, including:
- Linear: Two electron pairs arranged in a straight line (e.g., BeCl2).
- Trigonal Planar: Three electron pairs arranged in a plane (e.g., BF3).
- Tetrahedral: Four electron pairs arranged in a three-dimensional shape (e.g., CH4).
- Trigonal Bipyramidal: Five electron pairs with three in a plane and two above and below (e.g., PF5).
- Octahedral: Six electron pairs arranged in a three-dimensional shape (e.g., SF6).
Understanding Molecular Geometry
Molecular geometry, on the other hand, refers to the spatial arrangement of atoms in a molecule, considering only the bonding pairs of electrons. Unlike electron geometry, molecular geometry does not account for lone pairs of electrons. This distinction is crucial because lone pairs occupy more space than bonding pairs, affecting the overall shape of the molecule.
Molecular geometry can be determined by considering the number of bonding pairs and lone pairs around the central atom. Some common molecular geometries include:
- Linear: Two bonding pairs with no lone pairs (e.g., CO2).
- Trigonal Planar: Three bonding pairs with no lone pairs (e.g., SO3).
- Bent: Two bonding pairs and one or two lone pairs (e.g., SO2).
- Tetrahedral: Four bonding pairs with no lone pairs (e.g., CH4).
- Trigonal Pyramidal: Three bonding pairs and one lone pair (e.g., NH3).
Comparing Electron vs Molecular Geometry
To fully grasp the differences between Electron vs Molecular Geometry, let's compare them using a few examples:
| Molecule | Electron Geometry | Molecular Geometry |
|---|---|---|
| H2O | Tetrahedral | Bent |
| NH3 | Tetrahedral | Trigonal Pyramidal |
| BF3 | Trigonal Planar | Trigonal Planar |
| SF6 | Octahedral | Octahedral |
In the case of H2O, the electron geometry is tetrahedral because there are four electron pairs around the oxygen atom. However, the molecular geometry is bent because two of these pairs are lone pairs, which do not form bonds with other atoms.
For NH3, the electron geometry is also tetrahedral, but the molecular geometry is trigonal pyramidal due to the presence of one lone pair.
In BF3, both the electron and molecular geometries are trigonal planar because there are no lone pairs around the boron atom.
SF6 has an octahedral electron geometry and molecular geometry because all six electron pairs are bonding pairs.
💡 Note: The presence of lone pairs significantly affects the molecular geometry, making it different from the electron geometry.
Factors Affecting Electron vs Molecular Geometry
Several factors influence the Electron vs Molecular Geometry of a molecule:
- Number of Electron Pairs: The more electron pairs around the central atom, the more complex the geometry.
- Lone Pairs: Lone pairs occupy more space than bonding pairs, distorting the molecular geometry.
- Bond Angles: The angles between bonds can vary based on the presence of lone pairs and the type of hybridization.
- Hybridization: The hybridization of the central atom's orbitals (sp, sp2, sp3, etc.) affects the geometry.
Applications of Electron vs Molecular Geometry
Understanding Electron vs Molecular Geometry has numerous applications in chemistry and related fields:
- Predicting Molecular Properties: The shape of a molecule can influence its reactivity, polarity, and other properties.
- Drug Design: Knowing the geometry of molecules helps in designing drugs that can interact effectively with biological targets.
- Materials Science: The geometry of molecules can affect the properties of materials, such as their strength and conductivity.
- Catalysis: The shape of catalyst molecules can influence their effectiveness in chemical reactions.
For example, in drug design, understanding the molecular geometry of a drug molecule can help in predicting how it will interact with a receptor in the body. This knowledge is crucial for developing effective medications.
In materials science, the geometry of molecules can affect the properties of materials. For instance, the tetrahedral geometry of carbon atoms in diamond contributes to its exceptional hardness.
In catalysis, the geometry of catalyst molecules can influence their effectiveness. Catalysts with specific geometries can facilitate chemical reactions more efficiently.
💡 Note: The applications of electron and molecular geometry are vast and span across various scientific and industrial fields.
Examples of Electron vs Molecular Geometry
Let’s explore a few more examples to illustrate the differences between Electron vs Molecular Geometry:
Methane (CH4):
- Electron Geometry: Tetrahedral
- Molecular Geometry: Tetrahedral
- Explanation: All four electron pairs are bonding pairs, so the electron and molecular geometries are the same.
Ammonia (NH3):
- Electron Geometry: Tetrahedral
- Molecular Geometry: Trigonal Pyramidal
- Explanation: There are three bonding pairs and one lone pair, causing the molecular geometry to be trigonal pyramidal.
Water (H2O):
- Electron Geometry: Tetrahedral
- Molecular Geometry: Bent
- Explanation: There are two bonding pairs and two lone pairs, resulting in a bent molecular geometry.
Carbon Dioxide (CO2):
- Electron Geometry: Linear
- Molecular Geometry: Linear
- Explanation: There are two bonding pairs and no lone pairs, so the electron and molecular geometries are the same.
Sulfur Dioxide (SO2):
- Electron Geometry: Trigonal Planar
- Molecular Geometry: Bent
- Explanation: There are two bonding pairs and one lone pair, causing the molecular geometry to be bent.
These examples illustrate how the presence of lone pairs can significantly affect the molecular geometry, making it different from the electron geometry.
💡 Note: Understanding these examples can help in predicting the geometries of other molecules.
Visualizing Electron vs Molecular Geometry
Visualizing the geometries of molecules can be challenging, but there are several tools and methods that can help:
- Molecular Models: Physical models can be used to build and visualize the shapes of molecules.
- Computer Software: Software like ChemDraw, Avogadro, and others can generate 3D models of molecules.
- Ball-and-Stick Models: These models use balls to represent atoms and sticks to represent bonds, providing a clear visual representation of molecular geometry.
- Space-Filling Models: These models show the relative sizes of atoms and how they fit together in a molecule.
For instance, using a ball-and-stick model, you can clearly see the tetrahedral geometry of methane (CH4) and the trigonal pyramidal geometry of ammonia (NH3).
Space-filling models can help visualize how the atoms in a molecule occupy space, providing insights into the molecule's properties.
💡 Note: Visualizing molecular geometries can enhance understanding and make complex concepts more accessible.

This image provides a visual representation of different molecular geometries, helping to understand the spatial arrangement of atoms in molecules.

This image illustrates the electron geometries of various molecules, showing the arrangement of electron pairs around the central atom.
By comparing these visualizations, you can see how the presence of lone pairs affects the molecular geometry, making it different from the electron geometry.
Understanding the differences between Electron vs Molecular Geometry is essential for predicting the shapes and properties of molecules. This knowledge has wide-ranging applications in chemistry, materials science, drug design, and catalysis. By studying the spatial arrangement of electron pairs and atoms, we can gain insights into the behavior of molecules and their interactions with other substances.
In summary, electron geometry refers to the arrangement of electron pairs around a central atom, while molecular geometry refers to the arrangement of atoms in a molecule, considering only the bonding pairs. The presence of lone pairs significantly affects the molecular geometry, making it different from the electron geometry. Understanding these concepts is crucial for predicting molecular properties and has numerous applications in various scientific and industrial fields.
Related Terms:
- molecular geometry vs electron pair
- molecular geometry and electron chart
- electronic geometry vs molecular examples
- electron group chart
- molecular geometry chart
- electron domain vs molecular geometry