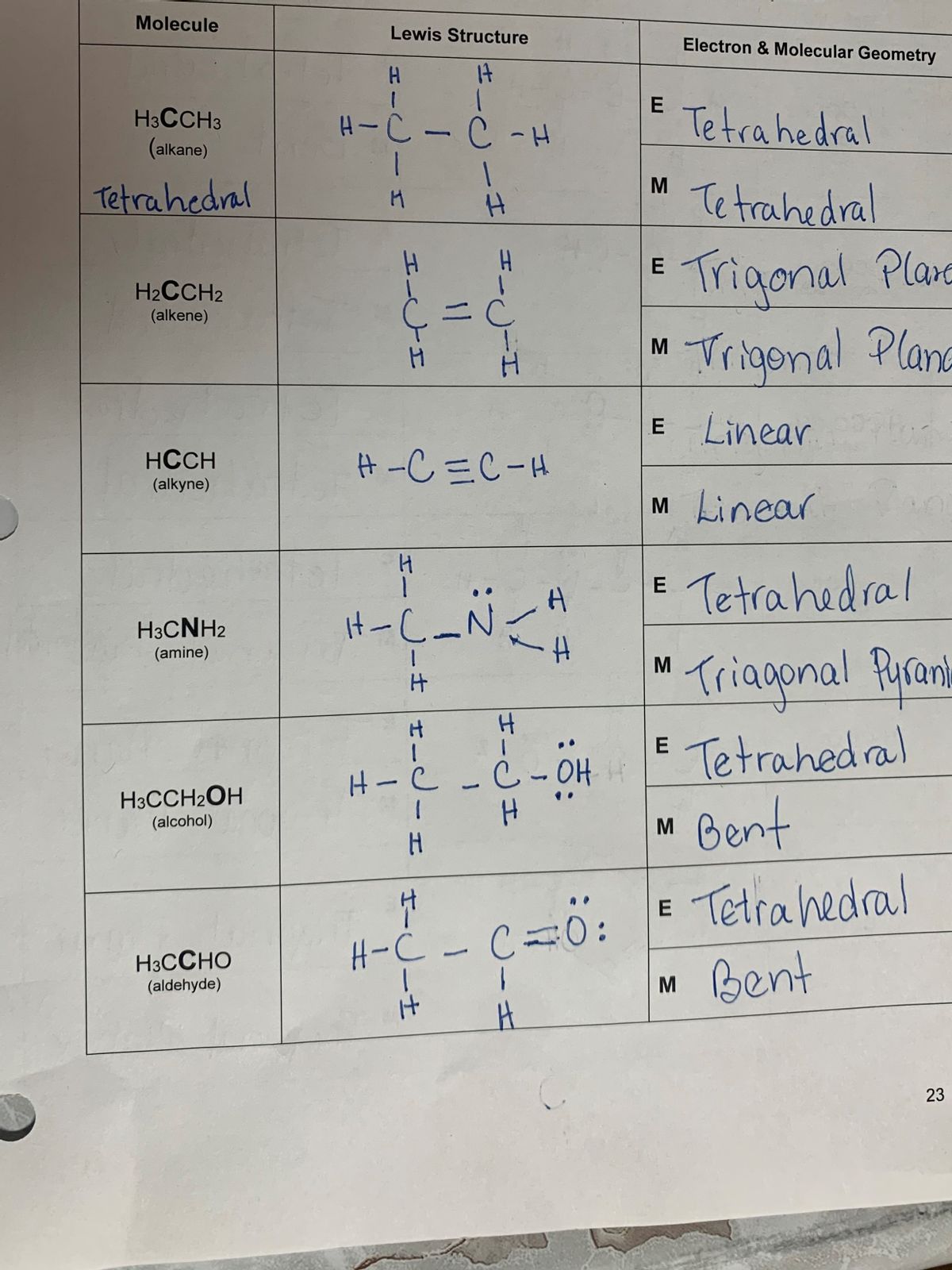
Understanding the Hcch Lewis Structure is fundamental for anyone studying chemistry, particularly organic chemistry. The Lewis structure, named after Gilbert N. Lewis, is a diagrammatic representation of the valence electrons in a molecule. It helps visualize the bonding between atoms and the lone pairs of electrons. This post will delve into the intricacies of the Hcch Lewis Structure, explaining its significance, how to draw it, and its applications in chemistry.
What is the Hcch Lewis Structure?
The Hcch Lewis Structure specifically refers to the Lewis structure of the methane molecule (CH₄). Methane is a simple hydrocarbon with one carbon atom bonded to four hydrogen atoms. The Lewis structure provides a clear picture of how these atoms are connected and how the electrons are distributed.
Drawing the Hcch Lewis Structure
Drawing the Hcch Lewis Structure involves several steps. Here’s a detailed guide:
Step 1: Determine the Total Number of Valence Electrons
First, identify the total number of valence electrons in the molecule. For methane (CH₄), carbon © has 4 valence electrons, and each hydrogen (H) has 1 valence electron. Therefore, the total number of valence electrons is:
4 (from C) + 4 × 1 (from H) = 8 valence electrons.
Step 2: Choose the Central Atom
In methane, the central atom is carbon © because it is the least electronegative and can form bonds with multiple hydrogen atoms.
Step 3: Connect the Atoms with Single Bonds
Draw single bonds between the central carbon atom and each hydrogen atom. This uses up 4 of the 8 valence electrons, leaving 4 electrons to be distributed.
Step 4: Distribute the Remaining Electrons
Place the remaining 4 electrons around the carbon atom to complete its octet. Since carbon has 4 valence electrons and needs 8 to complete its octet, the remaining 4 electrons will be placed as lone pairs around the carbon atom.
Step 5: Check for Octet Rule Compliance
Ensure that each atom has the correct number of electrons to satisfy the octet rule. In methane, carbon has 8 electrons (4 from bonds and 4 as lone pairs), and each hydrogen has 2 electrons (1 from the bond).
Importance of the Hcch Lewis Structure
The Hcch Lewis Structure is crucial for several reasons:
- Understanding Bonding: It helps in understanding the type of bonding (single, double, triple) between atoms.
- Predicting Molecular Shape: The Lewis structure can be used to predict the molecular geometry using the Valence Shell Electron Pair Repulsion (VSEPR) theory.
- Determining Polarity: It aids in determining the polarity of the molecule by showing the distribution of electrons.
- Reactivity: It provides insights into the reactivity of the molecule by showing the presence of lone pairs and bond types.
Applications of the Hcch Lewis Structure
The Hcch Lewis Structure has numerous applications in chemistry:
Organic Chemistry
In organic chemistry, the Lewis structure is used to understand the bonding in organic compounds, which is essential for predicting their properties and reactions.
Inorganic Chemistry
In inorganic chemistry, the Lewis structure helps in understanding the bonding in coordination compounds and complex ions.
Biochemistry
In biochemistry, the Lewis structure is used to understand the bonding in biomolecules like proteins, nucleic acids, and carbohydrates.
Material Science
In material science, the Lewis structure is used to understand the bonding in materials like semiconductors and polymers.
Common Mistakes to Avoid
When drawing the Hcch Lewis Structure, it’s important to avoid common mistakes:
- Incorrect Valence Electrons: Ensure you count the correct number of valence electrons for each atom.
- Incorrect Bonding: Make sure to draw the correct number of bonds between atoms.
- Ignoring Lone Pairs: Do not forget to include lone pairs of electrons around atoms that need them to complete their octet.
- Incorrect Central Atom: Choose the correct central atom based on electronegativity and bonding capacity.
🔍 Note: Always double-check your Lewis structure to ensure it complies with the octet rule and correctly represents the molecule's bonding and electron distribution.
Examples of Lewis Structures
Here are a few examples of Lewis structures for different molecules:
Water (H₂O)
Water has 2 hydrogen atoms and 1 oxygen atom. Oxygen has 6 valence electrons, and each hydrogen has 1 valence electron, totaling 8 valence electrons. The Lewis structure of water shows 2 single bonds between oxygen and hydrogen, with 2 lone pairs on the oxygen atom.
Ammonia (NH₃)
Ammonia has 1 nitrogen atom and 3 hydrogen atoms. Nitrogen has 5 valence electrons, and each hydrogen has 1 valence electron, totaling 8 valence electrons. The Lewis structure of ammonia shows 3 single bonds between nitrogen and hydrogen, with 1 lone pair on the nitrogen atom.
Carbon Dioxide (CO₂)
Carbon dioxide has 1 carbon atom and 2 oxygen atoms. Carbon has 4 valence electrons, and each oxygen has 6 valence electrons, totaling 16 valence electrons. The Lewis structure of carbon dioxide shows 2 double bonds between carbon and oxygen, with no lone pairs on the carbon atom.
Lewis Structures and Molecular Geometry
The Hcch Lewis Structure is closely related to molecular geometry. The Valence Shell Electron Pair Repulsion (VSEPR) theory uses the Lewis structure to predict the shape of a molecule. For example, methane (CH₄) has a tetrahedral geometry because the carbon atom is surrounded by four bonding pairs of electrons, which repel each other equally.
Lewis Structures and Polarity
The Hcch Lewis Structure also helps in determining the polarity of a molecule. Polarity depends on the distribution of electrons and the electronegativity of the atoms. For example, methane (CH₄) is non-polar because it has a symmetrical distribution of electrons, while water (H₂O) is polar because the oxygen atom is more electronegative than the hydrogen atoms, leading to an uneven distribution of electrons.
Lewis Structures and Reactivity
The Hcch Lewis Structure provides insights into the reactivity of a molecule. For example, the presence of lone pairs of electrons can indicate potential sites for chemical reactions. In ammonia (NH₃), the lone pair on the nitrogen atom can act as a Lewis base and donate electrons to form a coordinate covalent bond.
Lewis Structures and Resonance
Some molecules have multiple valid Lewis structures, a phenomenon known as resonance. Resonance structures are different Lewis structures of the same molecule that differ only in the position of electrons. For example, the ozone molecule (O₃) has two resonance structures, each showing a different arrangement of double bonds between the oxygen atoms.
Lewis Structures and Formal Charge
Formal charge is a concept used to determine the most stable Lewis structure of a molecule. It is calculated as the difference between the number of valence electrons in an isolated atom and the number of electrons assigned to that atom in the Lewis structure. The most stable Lewis structure is the one with the smallest formal charges.
Lewis Structures and Hybridization
Hybridization is the concept of mixing atomic orbitals to form new hybrid orbitals suitable for bonding. The Hcch Lewis Structure can help determine the hybridization of atoms in a molecule. For example, in methane (CH₄), the carbon atom is sp³ hybridized because it forms four single bonds with hydrogen atoms.
Lewis Structures and Bond Order
Bond order is the number of chemical bonds between a pair of atoms. The Hcch Lewis Structure can help determine the bond order in a molecule. For example, in carbon dioxide (CO₂), the bond order between carbon and oxygen is 2 because there are two double bonds between them.
Lewis Structures and Molecular Orbital Theory
Molecular Orbital (MO) theory is a more advanced theory that describes the distribution of electrons in molecules. It complements the Lewis structure by providing a more detailed picture of electron distribution and bonding. The Hcch Lewis Structure can be used as a starting point for MO theory calculations.
Lewis Structures and Electron Dot Diagrams
Electron dot diagrams are a simplified version of Lewis structures that show only the valence electrons as dots around the atomic symbols. They are useful for quickly visualizing the bonding and electron distribution in simple molecules. The Hcch Lewis Structure can be represented as an electron dot diagram for easier understanding.
Lewis Structures and Valence Bond Theory
Valence Bond (VB) theory is another theory that describes chemical bonding. It focuses on the overlap of atomic orbitals to form covalent bonds. The Hcch Lewis Structure can be used to understand the bonding in molecules according to VB theory.
Lewis Structures and Molecular Formula
The molecular formula of a compound provides the number of atoms of each element in the molecule. The Hcch Lewis Structure can be used to derive the molecular formula of a compound by counting the number of atoms of each element in the structure.
Lewis Structures and Structural Isomers
Structural isomers are compounds with the same molecular formula but different structures. The Hcch Lewis Structure can help identify structural isomers by showing the different arrangements of atoms and bonds in the molecule.
Lewis Structures and Functional Groups
Functional groups are specific groups of atoms within molecules that determine the characteristic chemical reactions of those molecules. The Hcch Lewis Structure can help identify functional groups in organic compounds by showing the bonding and electron distribution around the functional group.
Lewis Structures and Aromaticity
Aromaticity is a property of cyclic, planar molecules with a system of delocalized π electrons. The Hcch Lewis Structure can help determine the aromaticity of a molecule by showing the delocalization of π electrons in the ring structure.
Lewis Structures and Anti-Aromaticity
Anti-aromaticity is a property of cyclic, planar molecules with a system of delocalized π electrons that makes them unstable. The Hcch Lewis Structure can help determine the anti-aromaticity of a molecule by showing the delocalization of π electrons in the ring structure.
Lewis Structures and Non-Aromaticity
Non-aromaticity is a property of cyclic, planar molecules with a system of delocalized π electrons that do not exhibit aromatic or anti-aromatic properties. The Hcch Lewis Structure can help determine the non-aromaticity of a molecule by showing the delocalization of π electrons in the ring structure.
Lewis Structures and Hyperconjugation
Hyperconjugation is the interaction of the electrons in a σ bond (usually C-H or C-C) with an adjacent empty or partially filled p orbital or π orbital to give an extended molecular orbital. The Hcch Lewis Structure can help understand hyperconjugation by showing the delocalization of σ electrons.
Lewis Structures and Inductive Effects
The inductive effect is the polarization of a covalent bond due to the electronegativity difference between the bonded atoms. The Hcch Lewis Structure can help understand inductive effects by showing the distribution of electrons in the molecule.
Lewis Structures and Mesomeric Effects
The mesomeric effect is the delocalization of π electrons in a molecule due to the presence of electron-donating or electron-withdrawing groups. The Hcch Lewis Structure can help understand mesomeric effects by showing the delocalization of π electrons in the molecule.
Lewis Structures and Hydrogen Bonding
Hydrogen bonding is a type of intermolecular force that occurs when a hydrogen atom, bonded to a highly electronegative atom, experiences the electrostatic attraction of another highly electronegative atom nearby. The Hcch Lewis Structure can help understand hydrogen bonding by showing the distribution of electrons in the molecule.
Lewis Structures and Dipole-Dipole Interactions
Dipole-dipole interactions are intermolecular forces that occur between polar molecules. The Hcch Lewis Structure can help understand dipole-dipole interactions by showing the distribution of electrons in the molecule and the resulting polarity.
Lewis Structures and London Dispersion Forces
London dispersion forces are weak intermolecular forces that occur due to the instantaneous dipole moments in molecules. The Hcch Lewis Structure can help understand London dispersion forces by showing the distribution of electrons in the molecule.
Lewis Structures and Ionic Bonding
Ionic bonding occurs when one or more electrons are transferred from one atom to another, forming ions that are held together by electrostatic forces. The Hcch Lewis Structure can help understand ionic bonding by showing the transfer of electrons between atoms.
Lewis Structures and Metallic Bonding
Metallic bonding occurs in metals, where the valence electrons are delocalized and form a “sea” of electrons that hold the positively charged metal ions together. The Hcch Lewis Structure can help understand metallic bonding by showing the delocalization of valence electrons in the metal.
Lewis Structures and Coordinate Covalent Bonding
Coordinate covalent bonding occurs when one atom donates a pair of electrons to another atom to form a covalent bond. The Hcch Lewis Structure can help understand coordinate covalent bonding by showing the donation of electron pairs between atoms.
Lewis Structures and Three-Center Two-Electron Bonds
A three-center two-electron bond is a type of chemical bond where two electrons are shared among three atoms. The Hcch Lewis Structure can help understand three-center two-electron bonds by showing the distribution of electrons among the three atoms.
Lewis Structures and Four-Center Two-Electron Bonds
A four-center two-electron bond is a type of chemical bond where two electrons are shared among four atoms. The Hcch Lewis Structure can help understand four-center two-electron bonds by showing the distribution of electrons among the four atoms.
Lewis Structures and Five-Center Two-Electron Bonds
A five-center two-electron bond is a type of chemical bond where two electrons are shared among five atoms. The Hcch Lewis Structure can help understand five-center two-electron bonds by showing the distribution of electrons among the five atoms.
Lewis Structures and Six-Center Two-Electron Bonds
A six-center two-electron bond is a type of chemical bond where two electrons are shared among six atoms. The Hcch Lewis Structure can help understand six-center two-electron bonds by showing the distribution of electrons among the six atoms.
Lewis Structures and Seven-Center Two-Electron Bonds
A seven-center two-electron bond is a type of chemical bond where two electrons are shared among seven atoms. The Hcch Lewis Structure can help understand seven-center two-electron bonds by showing the distribution of electrons among the seven atoms.
Lewis Structures and Eight-Center Two-Electron Bonds
An eight-center two-electron bond is a type of chemical bond where two electrons are shared among eight atoms. The Hcch Lewis Structure can help understand eight-center two-electron bonds by showing the distribution of electrons among the eight atoms.
Lewis Structures and Nine-Center Two-Electron Bonds
A nine-center two-electron bond is a type of chemical bond where two electrons are shared among nine atoms. The Hcch Lewis Structure can help understand nine-center two-electron bonds by showing the distribution of electrons among the nine atoms.
Lewis Structures and Ten-Center Two-Electron Bonds
A ten-center two-electron bond is a type of chemical bond where two electrons are shared among ten atoms. The Hcch Lewis Structure can help understand ten-center two-electron bonds by showing the distribution of electrons among the ten atoms.
Lewis Structures and Eleven-Center Two-Electron Bonds
An eleven-center two-electron bond is a type of chemical bond where two electrons are shared among eleven atoms. The Hcch Lewis Structure can help understand eleven-center two-electron bonds by showing the distribution of electrons among the eleven atoms.
Lewis Structures and Twelve-Center Two-Electron Bonds
A twelve-center two-electron bond is a type of chemical bond where two electrons are shared among twelve atoms. The Hcch Lewis Structure can help understand twelve-center two-electron bonds by showing the distribution of electrons among the twelve atoms.
Lewis Structures and Thirteen-Center Two-Electron Bonds
A thirteen-center two-electron bond is a type of chemical bond where two electrons are shared among thirteen atoms. The Hcch Lewis Structure can help understand thirteen-center two-electron bonds by showing the distribution of electrons among the thirteen atoms.
Lewis Structures and Fourteen-Center Two-Electron Bonds
A fourteen-center two-electron bond is a type of chemical bond where two electrons are shared among fourteen atoms. The Hcch Lewis Structure can help understand fourteen-center two-electron bonds by showing the distribution of electrons among the fourteen atoms.
Lewis Structures and Fifteen-Center Two-Electron Bonds
A fifteen-center two-electron bond is a type of chemical bond where two electrons are shared among fifteen atoms. The Hcch Lewis Structure can help understand fifteen-center two-electron bonds by showing the distribution of electrons among the fifteen atoms.
Lewis Structures and Sixteen-Center Two-Electron Bonds
A sixteen-center two-electron bond is a type of chemical bond where two electrons are shared among sixteen atoms. The Hcch Lewis Structure can help understand sixteen-center two-electron bonds by showing the distribution of electrons among the sixteen atoms.
Lewis Structures and Seventeen-Center Two-Electron Bonds
A seventeen-center two-electron bond is a type of chemical bond where two electrons are shared among seventeen atoms. The Hcch Lewis Structure can help understand seventeen-center two-electron bonds by showing the distribution of electrons among the seventeen atoms.
Lewis Structures and Eighteen-Center Two-Electron Bonds
An eighteen-center two-electron bond is a type of chemical bond where two electrons are shared among eighteen atoms. The Hcch Lewis Structure can help understand eighteen-center two-electron bonds by showing the distribution of electrons among the eighteen atoms.
Lewis Structures and Nineteen-Center
Related Terms:
- hcch molecule
- c2h2 lewis structure
- c2h2 lewis structure electron geometry
- hcch lewis structure geometry
- lewis structure of h2c ch2
- ch3 lewis structure