Understanding the periodic trends of elements is fundamental to grasping the behavior of atoms and molecules. One of the key trends that chemists and students alike must comprehend is the Nuclear Charge Trend. This trend refers to the change in the nuclear charge (the number of protons in the nucleus) as you move across a period or down a group in the periodic table. The nuclear charge directly influences the chemical properties of elements, making it a crucial concept in chemistry.
What is Nuclear Charge?
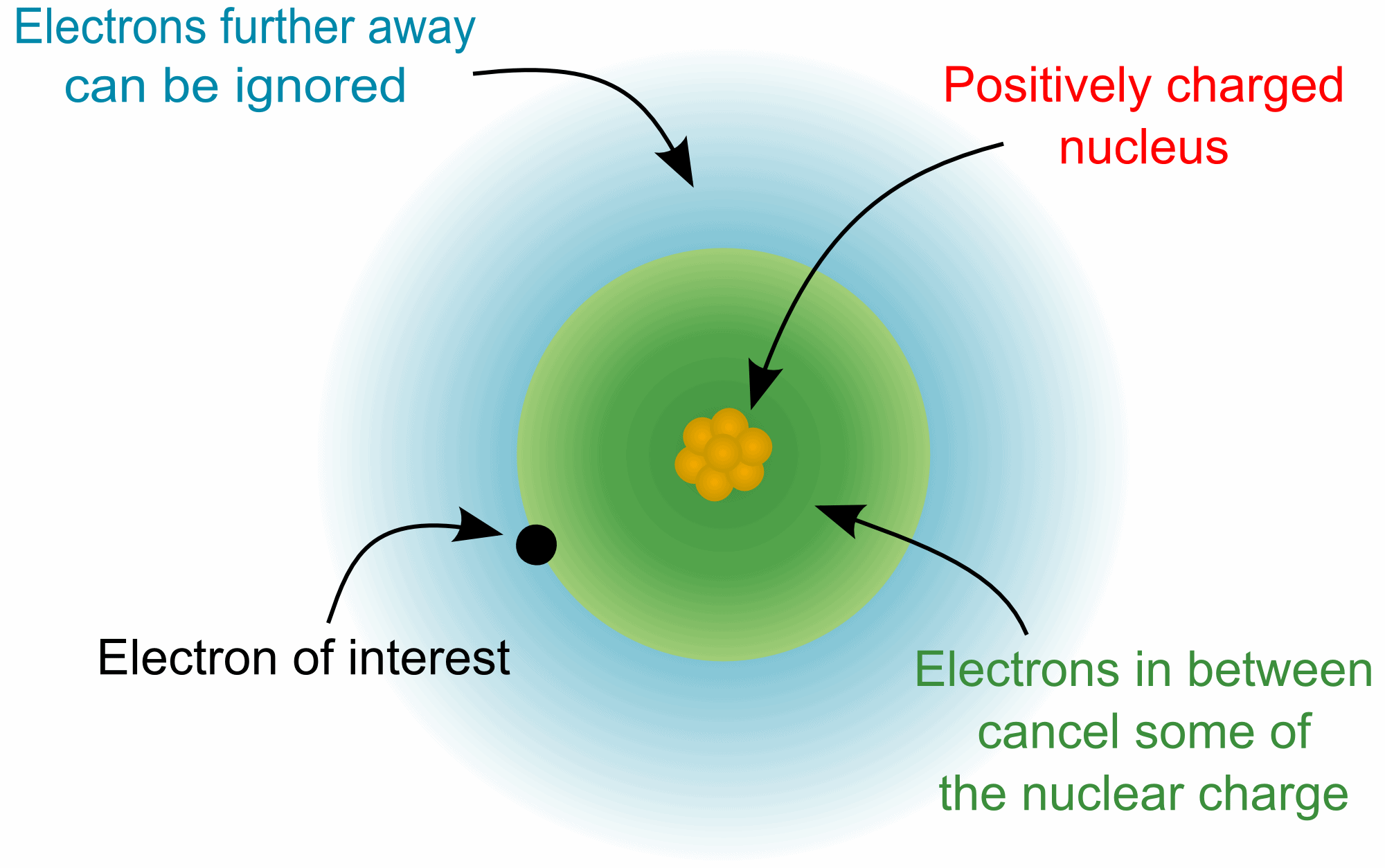
The nuclear charge of an atom is determined by the number of protons in its nucleus. Since protons are positively charged, the nuclear charge is essentially the positive charge of the nucleus. This charge is balanced by the negatively charged electrons orbiting the nucleus. The nuclear charge is a critical factor in determining the element’s atomic number and its position in the periodic table.
Nuclear Charge Trend Across a Period
As you move from left to right across a period in the periodic table, the nuclear charge increases. This is because each subsequent element has one more proton than the previous one. For example, in the second period, lithium (Li) has a nuclear charge of +3, beryllium (Be) has +4, boron (B) has +5, and so on, up to neon (Ne) with a nuclear charge of +10.
This increase in nuclear charge has several implications:
- Increasing Effective Nuclear Charge: As the nuclear charge increases, the effective nuclear charge (the net positive charge experienced by an electron) also increases. This is because the additional protons in the nucleus attract the electrons more strongly.
- Decreasing Atomic Radius: The stronger attraction between the nucleus and the electrons pulls the electrons closer to the nucleus, resulting in a smaller atomic radius.
- Increasing Ionization Energy: The increased nuclear charge makes it harder to remove an electron from the atom, leading to higher ionization energies.
- Decreasing Electronegativity: Electronegativity generally increases across a period due to the increasing nuclear charge, making it easier for the atom to attract electrons in a chemical bond.
Nuclear Charge Trend Down a Group
When moving down a group in the periodic table, the nuclear charge also increases, but the trend is less straightforward than across a period. Each element in a group has one more proton and one more electron shell than the element above it. This additional electron shell shields the outer electrons from the increased nuclear charge, mitigating its effects.
Key points to consider:
- Increasing Atomic Radius: Despite the increase in nuclear charge, the addition of new electron shells causes the atomic radius to increase. The outer electrons are farther from the nucleus, leading to a larger atomic radius.
- Decreasing Ionization Energy: The shielding effect of the additional electron shells reduces the effective nuclear charge experienced by the outer electrons, making it easier to remove an electron. Thus, ionization energy decreases down a group.
- Decreasing Electronegativity: The increased distance between the nucleus and the outer electrons, combined with the shielding effect, results in a decrease in electronegativity down a group.
Impact of Nuclear Charge on Chemical Properties
The Nuclear Charge Trend significantly influences the chemical properties of elements. For instance, elements with higher nuclear charges tend to be more reactive because they have a stronger attraction for electrons. This is why metals on the left side of the periodic table (with lower nuclear charges) are generally more reactive than nonmetals on the right side (with higher nuclear charges).
Additionally, the nuclear charge affects the types of bonds elements can form. Elements with higher nuclear charges are more likely to form ionic bonds, where electrons are transferred from one atom to another, while elements with lower nuclear charges are more likely to form covalent bonds, where electrons are shared between atoms.
Examples of Nuclear Charge Trend
To illustrate the Nuclear Charge Trend, let’s consider a few examples:
In the third period, the nuclear charge increases from sodium (Na) with +11 to argon (Ar) with +18. This increase results in a decrease in atomic radius and an increase in ionization energy and electronegativity.
In Group 1 (alkali metals), the nuclear charge increases from lithium (Li) with +3 to francium (Fr) with +87. However, the atomic radius increases, and the ionization energy and electronegativity decrease due to the shielding effect of the additional electron shells.
Periodic Table and Nuclear Charge
The periodic table is a visual representation of the Nuclear Charge Trend. Elements are arranged in order of increasing atomic number, which corresponds to the number of protons (and thus the nuclear charge) in the nucleus. This arrangement highlights the periodic trends in atomic radius, ionization energy, and electronegativity.
Here is a simplified table showing the nuclear charge for the first 18 elements:
| Element | Symbol | Nuclear Charge |
|---|---|---|
| Hydrogen | H | +1 |
| Helium | He | +2 |
| Lithium | Li | +3 |
| Beryllium | Be | +4 |
| Boron | B | +5 |
| Carbon | C | +6 |
| Nitrogen | N | +7 |
| Oxygen | O | +8 |
| Fluorine | F | +9 |
| Neon | Ne | +10 |
| Sodium | Na | +11 |
| Magnesium | Mg | +12 |
| Aluminum | Al | +13 |
| Silicon | Si | +14 |
| Phosphorus | P | +15 |
| Sulfur | S | +16 |
| Chlorine | Cl | +17 |
| Argon | Ar | +18 |
📝 Note: The nuclear charge is equal to the atomic number of the element, which is the number of protons in the nucleus.
Applications of Nuclear Charge Trend
The understanding of the Nuclear Charge Trend has numerous applications in chemistry and related fields. For example, it helps in predicting the reactivity of elements, designing new materials, and understanding chemical reactions. In industrial chemistry, knowledge of nuclear charge trends is crucial for optimizing processes and ensuring the safety and efficiency of chemical reactions.
In environmental science, the Nuclear Charge Trend aids in understanding the behavior of pollutants and contaminants. Elements with higher nuclear charges may form more stable compounds, affecting their mobility and toxicity in the environment.
In medicine, the trend is used in the development of pharmaceuticals. The nuclear charge of an element can influence its interaction with biological molecules, affecting drug efficacy and safety.
In summary, the Nuclear Charge Trend is a fundamental concept in chemistry that helps explain the periodic trends in atomic properties. Understanding this trend is essential for predicting the behavior of elements and their compounds, designing new materials, and optimizing chemical processes. By grasping the Nuclear Charge Trend, students and professionals can gain a deeper insight into the periodic table and its applications in various fields.
Related Terms:
- effective nuclear charge chart
- effective nuclear charge
- nuclear effective charge trend
- effective nuclear charge definition
- periodic table with nuclear charges
- nuclear charge across a period